WELCOME TO THE LAST BLOG!
In today’s final blog, we’ll be discussing tree diversity!
I can’t stop thinking of Christmas trees! I smell Pine everywhere I go!
For the lab, we actually went off campus! We traveled to a trail about 10 minutes out. When we got there, the first thing I noticed was how wet the ground was. If I remember correctly, it rained a couple days before the lab. It was also really cold outside with some occasional winds. No one could feel their toes after about 30 minutes outside! Other than being a frequently used trail, there were no other obvious signs of disturbance. I’m sure the forest could have been a little more dense if they put fences along the trail. It was also difficult to tell since the leaves have fallen off the trees and a lot of dead plant material remained on the bed. All the trees were relatively small. However, this could’ve simply been the area we chose. Another part of the trail seemed to have way bigger trees.
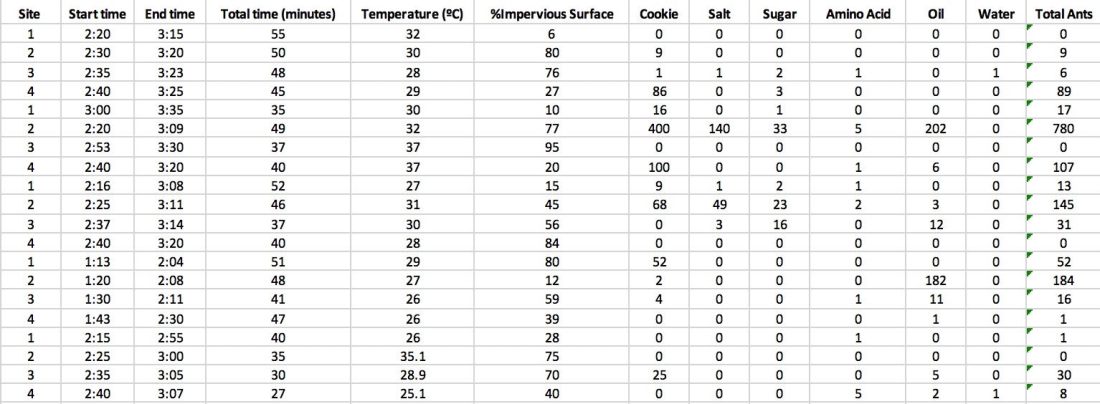
The goal of the lab was to observe species richness. In groups, we did this by using transects. There are two types of transects: Line transect and belt transect. A line transect, which is what we used, is when samples are taken along a straight line on either side of the transect at certain intervals. Belt transect is similar but involves quadrats. Instead of just using a line and counting the samples along the line, quadrats are used to get a bigger sample of data. This process takes longer but gives more complete data. However, for time’s sake, we simply used the line transect.
We started by finding a good spot along the trail that wasn’t too dense with trees, but not too sparse either. We just had to be able to walk through relatively easily. When we found a spot, we measured out 50 meters (165 feet) using a transect tape into the interior of the forest. Once that was laid out, we flipped a coin to find out which side of the transect line we were measuring on. For my group, heads was to the right and tails was to the left. Once the direction was figured out, we would use a 1.5 meter (5 foot) string and taut the strong out from the transect line. We would then proceed to count every different tree species that touched the taut string at every 1.5 meter (5 foot) interval.
Disclaimer: The worksheet we used had some typos in it and didn’t record the line transect at 75 and 95 feet. That’s one error we might want to consider when looking at our data. I don’t think it would be a massive difference but everything counts.
Here were our results!
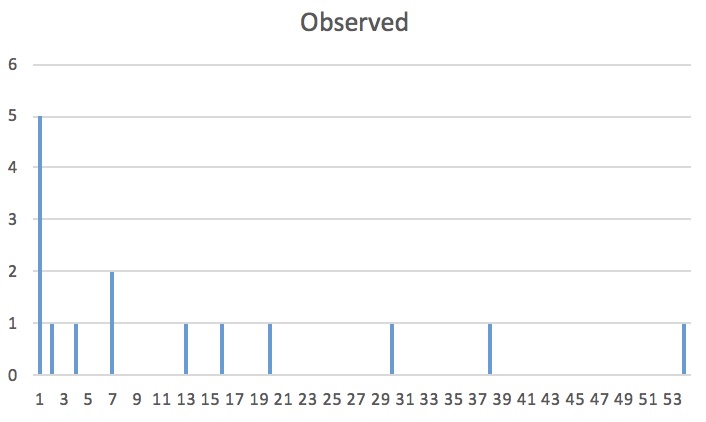
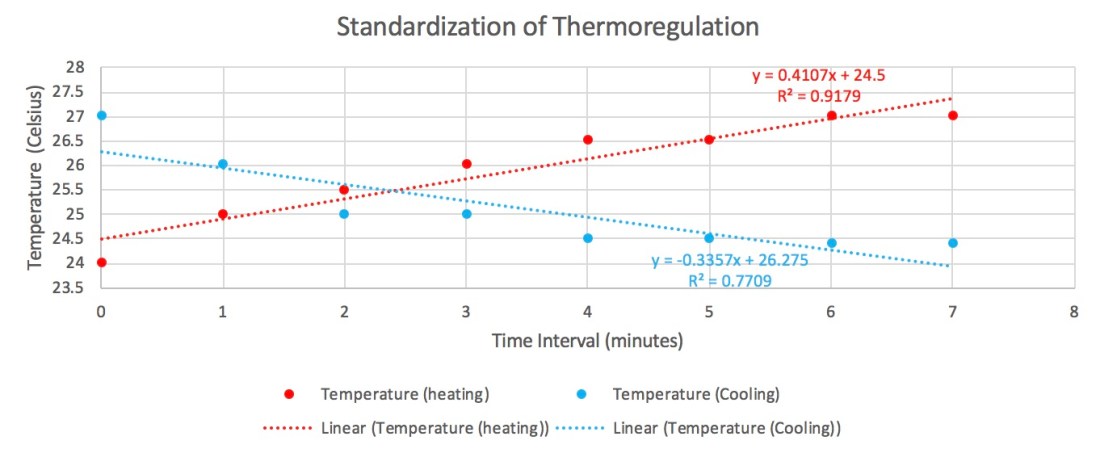
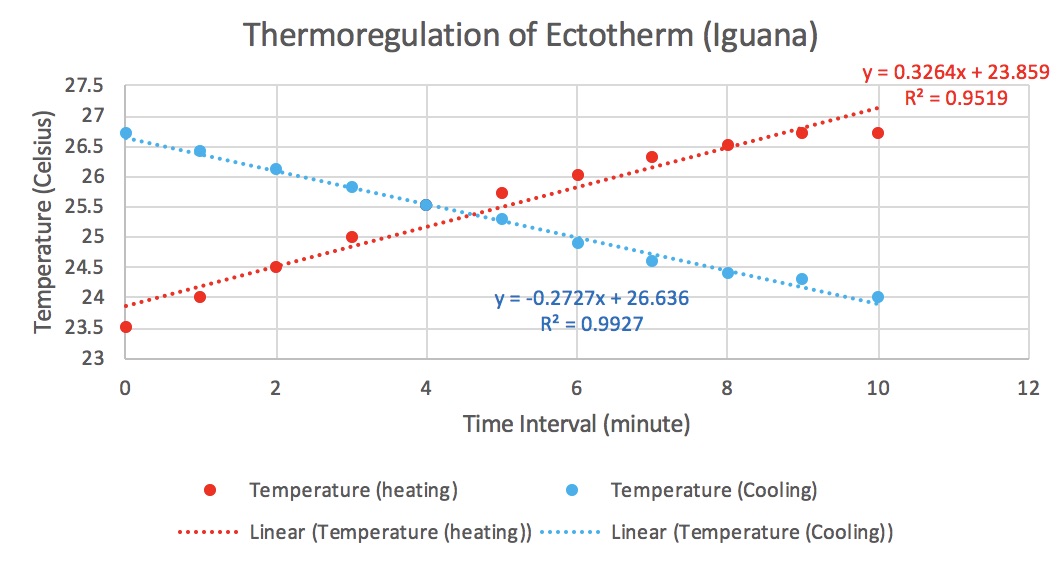
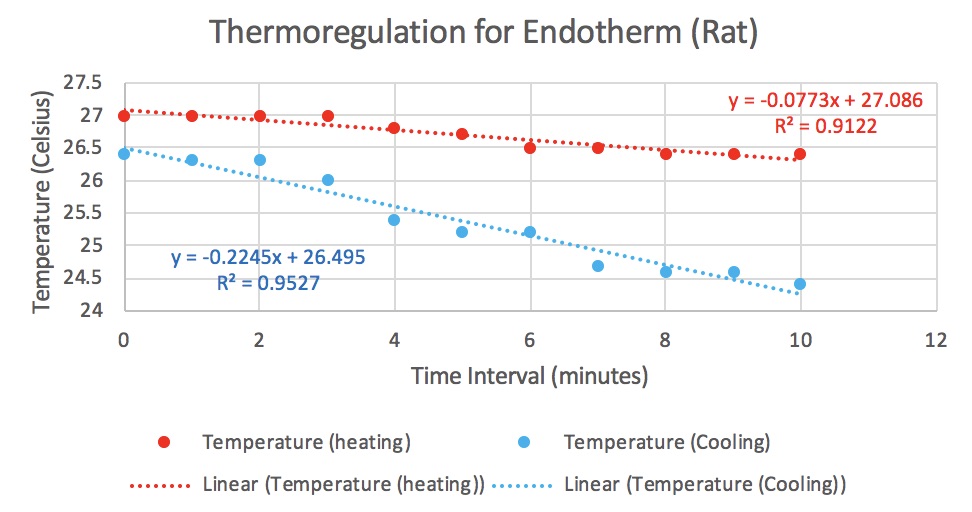
As you can tell from our scatter plot, there is a slight positive correlation when comparing number of tree species to the line transect.
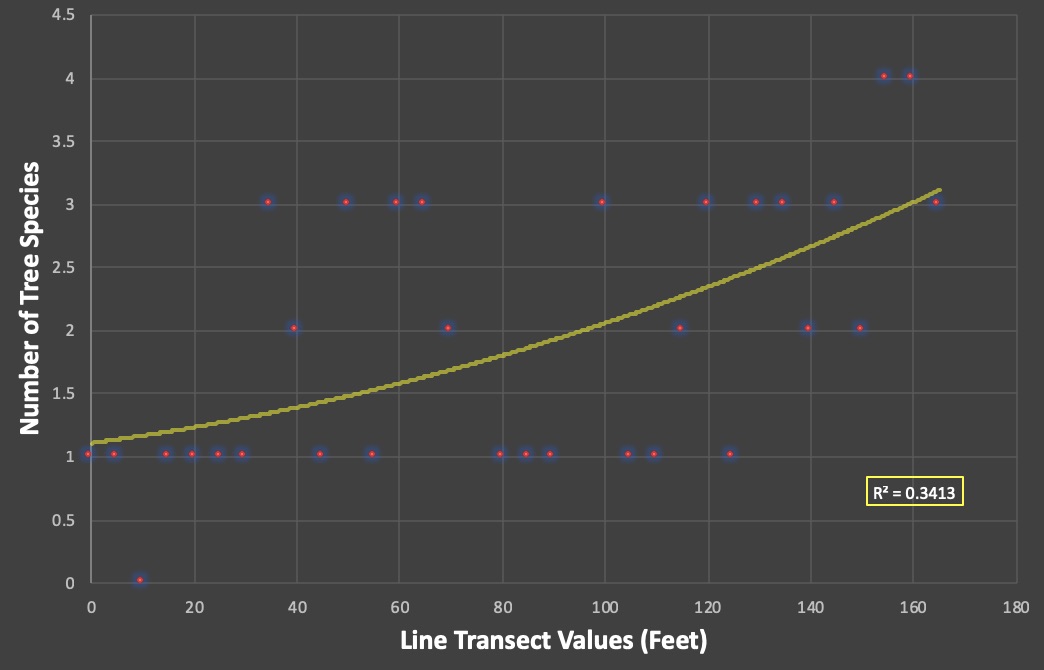
This means that the farther we move into the interior of the forest, the more species we see. Because the r-squared value is 0.3413, this shows us that the pattern is weaker than it is strong but it is not absurdly weak. To better understand this, we needed to run a regression analysis. We only need to pay attention to the p-value.

The p-value is below 0.05 which means it is statistically significant!
Habitat fragmentation greatly effects species diversity. According to a peer reviewed article called Habitat fragmentation, Tree diversity, and Plant Invasion Interact to Structure Forest Caterpillar Communities by John O. Stireman III, et al., “habitat fragmentation and invasive species are two of the most prominent threats to terrestrial ecosystems”. A lack of species diversity causes more problems than you think. Because there aren’t as many species of trees available, this means that there can’t be a very diverse number of organisms either. In Stireman III’s study, they noticed that when there was an abundance of Honeysuckle, caterpillar numbers and diversity would decrease (Stireman III).
Here’s another example of the effects of habitat fragmentation:
There’s an article called Gene Flow Halted by Fragmented Forests by Asian Scientist Newsroom that discusses the endangered maple tree and how it has been affected by habitat fragmentation. According to the scientists, they believe that the conservation of river floodplain ecosystems could maintain the genetic diversity of these maple trees. Because of this, these maple trees are important as reservoirs of genetic diversity and therefore should be conserved. When the scientists conducted an experiment comparing young and old maple trees, they noticed that the small, young trees had a higher level of genetic differentiation compared to the older maple trees. This leads us to a bigger picture… What is helping promote diversity? Well, if there is a river somewhere, the water, carrying all sorts of nutrients, minerals, and organisms helps out more than one can imagine. This is why many people are trying to preserve forests along rivers. This gives this area an advantage and promotes genetic diversity that other forest patches would not have.
Overall, tree diversity is such an important subject that requires patience and a lot of knowledge. Let’s just say that you need a lot of diverse information. Ha, get it? Ok, i’ll show myself out now…
Anyway, this is the last blog! Thank you for being part of the blog family! Hope you enjoyed everything you read!

-Louanne Maes
Peer Review Citations
Stireman, John O., et al. “Habitat Fragmentation, Tree Diversity, and Plant Invasion Interact to Structure Forest Caterpillar Communities.” Oecologia, vol. 176, no. 1, 2014, pp. 207–224.


 Welcome Back
Welcome Back